Neon is the first p-block noble gas and the first element with a true octet of electrons. Number of orbit in Neon 2.
Electron Configurations For The Third And Fourth Periods Video Khan Academy
Answer verified by Toppr.
Electron configuration of neon meaning. Since in 2nd orbit the maximum number of electrons is equal to 8 and there are 11 electrons in sodium atom thus the eleventh electron will go in third orbit. An electronic configuration is the way in which electrons. Z eff F 9 2 7.
Are arranged in an atom. Which has the same electron configuration as the neon atom. N a B.
The remaining six electrons will go in the 2p orbital. Ne has 201797 atomic mass 10 electrons and protons 101797 neutrons electron configuration is He2S22p6. Electron Configuration and Oxidation States of Neon Electron configuration of Neon is He 2s2 2p6.
The term noble gas electron configuration indicates that all atomic orbitals are completely filled with electrons. Therefore number of electrons 11. The atomic number for F is 9 therefore.
Neon is the tenth element with a total of 10 electrons. Possible oxidation states are 0. Electron configuration of Neon is He 2s2 2p6.
Because electrons in filled inner orbitals are closer to the nucleus and more tightly bound to it they are rarely involved in chemical reactions. 119 rows 6142015 ELECTRON CONFIGURATION. We could also have written used He as our starting point as it is the closest noble gas to Neon and continued the configuration from.
6122017 Coincidently Neon itself is a noble gas so we could also write the electron configuration as Ne. 11182020 The term electron configuration refers to the electron sequence or the order of the electrons present in an atom of a particular chemical element. 11132020 Neon is a chemical element with atomic number 10 which means there are 10 protons and 10 electrons in the atomic structure.
Thus electronic configuration of neon is. Atomic number of sodium 11. Our Periodic Element comparison tool allows you to compare Periodic Elements properties side by side for all 118 elements SchoolMyKids Interactive Dynamic Periodic Table Periodic Table Element Comparison tool Element Property trends.
Electrons in shells Different shells can hold different maximum numbers of electrons. Neon symbol is Ne atomic number 10 that locate in 2 period of noble gas group. All elements contain some specified number of electrons which is identical to the atomic number of that element.
The element is incredibly non- reactive because of its electron configuration. The chemical symbol for Neon is Ne. Moreover what is the electron configuration for neon.
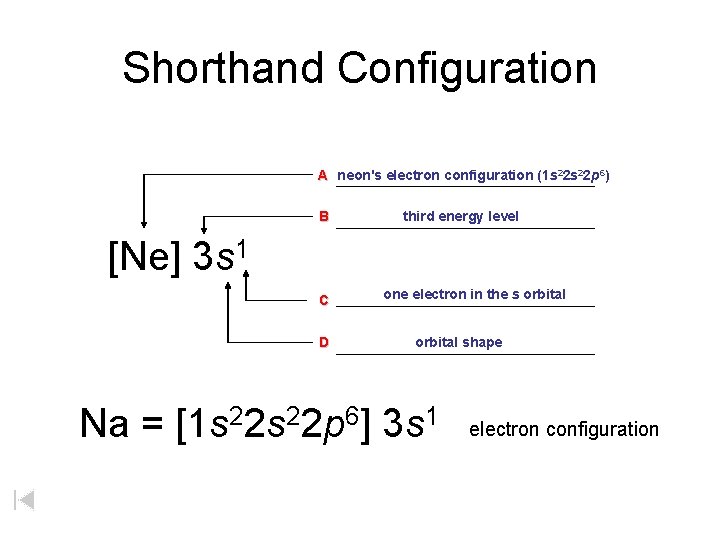
Neon textNe has an atomic number of 10 meaning that a neutral atom also has 10 electrons. Write the full electron configuration for neon Ne. 6152017 For example Ne represents the 1 s2 2 s2 2 p6 electron configuration of neon Z 10 so the electron configuration of sodium with Z 11 which is 1 s2 2 s2 2 p6 3 s1 is written as Ne3 s1.
The electron configuration is the same as for neon and the number of nonvalence electrons is 2. Click to see full answer. Since 1s can only hold two electrons the next 2 electrons for Ne go in the 2s orbital.
Electronic configuration of Sodium. Thus electronic configuration of sodium is. In writing the electron configuration for neon the first two electrons will go in the 1s orbital.
Therefore the Ne electron configuration will be 1s 2 2s 2 2p 6. N a As N a Atomic number 10 and N e Atomic number 10have same number of electrons therefore their electronic configuration will also be same. Correct option is.
The first 2 electrons are found in the first energy level and. 982020 When we reach neon with Z 10 we have filled the 2p subshell giving a 1s 2 2s 2 2p 6 electron configuration. Notice that for neon as for helium all the orbitals through the 2 p level are completely filled.
Compare Neon vs Krypton of the Periodic Table on all their Facts Electronic Configuration Chemical Physical Atomic properties.
Electron Configuration For Neon Ne
4 Ways To Write Electron Configurations For Atoms Of Any Element
Electron Configuration Na 1 S 2 2 P
Electron Configurations For The Second Period Video Khan Academy
Electron Configuration For Boron Learn Lif Co Id
Electron Configuration Elements 1 20 Good Science
Building Up The Periodic Table
Electron Configuration Detailed Explanation With Examples
Neon Electron Configuration Electron Configuration Electrons Configuration
Electron Configurations How To Write Out The S P D F Electronic Arrangements Of Atoms Ions Periodic Table Oxidation States Using Orbital Notation Gce A Level Revision Notes
Electronic Structure Of Atoms Electron Configurations Chemistry Atoms First
What Is Electronic Configuration Quora
Electron Configuration Thecuriousastronomer
Neon Protons Neutrons Electrons Electron Configuration
Electron Configuration And Periodic Trends Ppt Video Online Download
How To Write A Noble Gas Configuration For Atoms Of An Element
Electron Configuration Paulis Exclusion Principle Pauli Exclusion Principle
Electron Configuration Fillingorder Of Electrons In An Atom
